
Mahl- und Dispersionstechnik
Scheiben-Rührwerkskugelmühle
DYNO-MILL ECO 5
Die ideale Scheibenrührwerkskugelmühle für Kleinproduktionen
Branchen
Praktisch und wirtschaftlich für Kleinproduktionen
Die praktische Rührwerkskugelmühle für Kleinproduktionen wird für das kontinuierliche Dispergieren und Nassmahlen von dünnflüssigen bis viskosen, pumpfähigen Produkten eingesetzt.
Funktionsweise
Die DYNO-MILL ECO 5 ist mit den traditionellen KD-Rührscheiben ausgerüstet. Über auf der Rührwelle montierte, leicht auswechselbare Rührscheiben wird die für die Dispergierung und Mahlung notwendige Energie auf die Mahlkörper übertragen. Das zu verarbeitende Produkt wird der Mühle über eine externe Förderpumpe zugeführt. Der Rückhalt der Mahlkörper in der Mühle erfolgt mittel eines dynamischen Spaltseparators. Die Spaltbreite wird mit Abstandsnormteilen entsprechend der Mahlkörpergrössen eingestellt. Es können Mahlkörper aus verschiedensten Werkstoffen und Grössen (0.5-2.5 mm) eingesetzt werden.
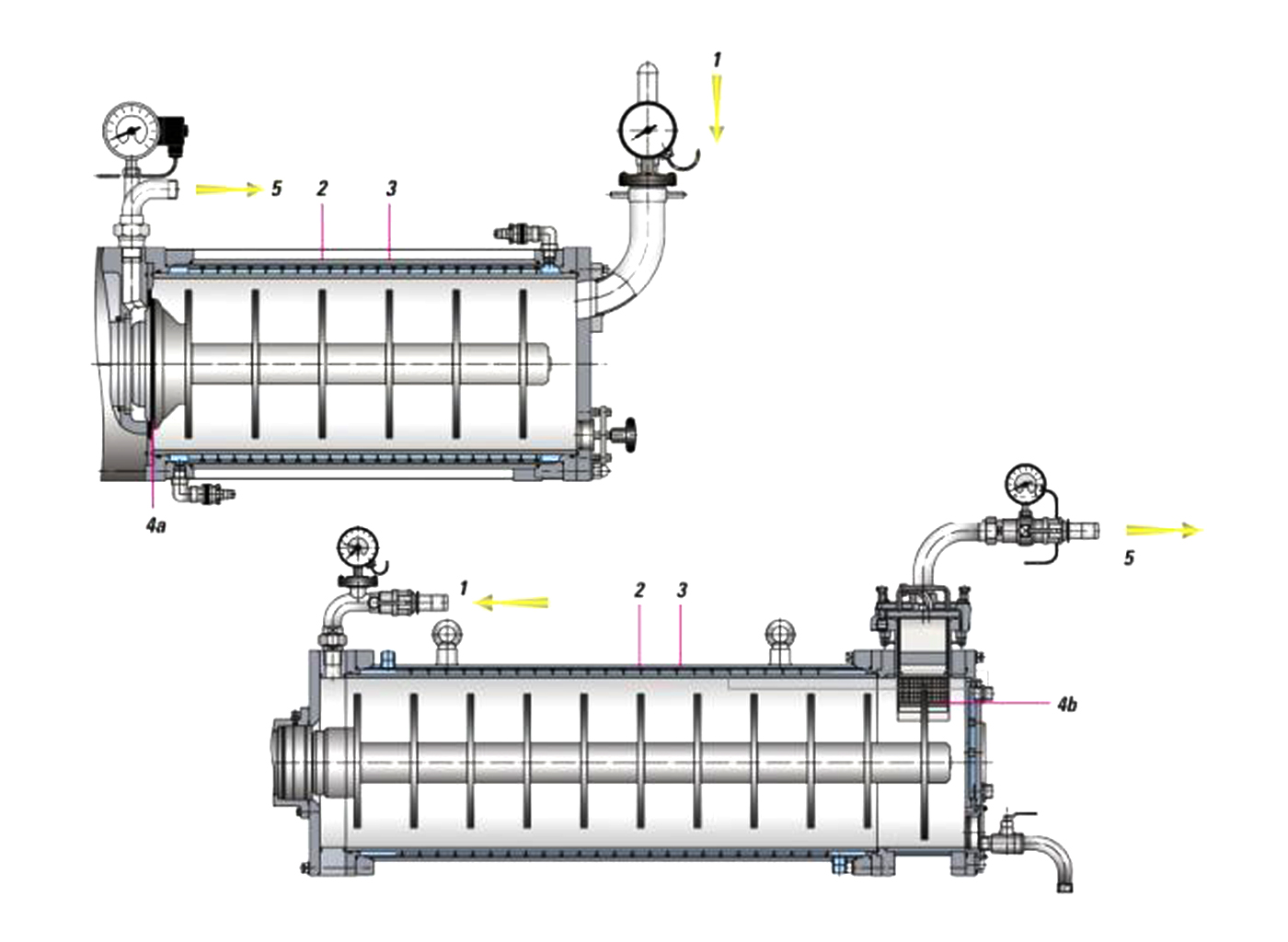
Bewährtes Mahlkammersystem
- Produkteinlauf/Mahlkörpereinfüllung
- DYNO-Rührscheiben
- Kühlbarer Mahlbehälter mit leicht auswechselbarem Mahlzylinder
- Dynamischer Spaltseparator
- Produktauslauf
Eigenschaften
- Ideal für Passagenfahrweise mit gleichbleibender Effizienz und zum Erreichen engster Kornspektren im Mikronbereich
- Für jedes Produkt die ideale Ausrüstung mit der Auswahl verschiedenster Materialien für die produktberührenden Bauteile
- Kleines Prozessvolumen und ideale Kühleigenschaften mit Zwangsführung des Kühlwassers im Aussenmantel erlauben das Verarbeiten von temperaturempfindlichen Produkten
- Einfaches Handling – mit wenigen Handgriffen lässt sich die DYNO-MILL ECO 5 demontieren und ein schnelles Umrüsten auf eine andere Mühlenkonfiguration ist gewährleistet
- Einfach Bedienung und Einstellung der Betriebsparameter
- Durch gleichbleibendes Mahlkonzept ist der Scale-up auf alle Produktionsmühlen der DYNO-MILL KD Baureihe möglich
- Niedrige Betriebs- und Wartungskosten

Download
Finden Sie weitere Informationen zu unserer Scheibenrührwerkskugelmühle in kompakter Form als PDF-Datei. Praktisch und wirtschaftlich für Kleinproduktionen.
Technische Spezifikationen
| Typ | ECO 5 |
|---|---|
| Mahlbehältervolumen (l) | 5 |
| Leistungsbereich (l/h) | 20-200 |
| Antrieb Mühle (kW) | 10 |
| Kühlwasserbedarf (l/h) | 800-1000 |
| Gewicht (kg) | 250-300 |
| Abmessung L x B x H (mm) | 1001 x 797 x 654 |
Wir sind für Sie da
Mit unseren lokalen Standorten und Fachspezialisten vor Ort stehen wir Ihnen gerne als kompetentes Maschinenbauunternehmen und bewährter Prozessexperte zur Verfügung.
„*“ zeigt erforderliche Felder an








